Publications
Presentations/Lectures
I worked on signal transduction pathways, more precisely the MAPK pathway, and virology, Theiler's murine encephalomyelitis virus and BK virus.
In the menu list above I provide an overview of oral (presentations, lectures, research visits) and written (original research, reviews) work.
Below is a little piece of text that summarizes what I did during my PhD.
Study of the MAPKAPK MK5
Cells communicate with one another by exchanging signals. Each particular signal (e.g. growth hormones), triggers specific responses (e.g. cell division). Since a cell receives several signals at once and each signal activates a particular cascade of events (signalling pathway), the response of the cell depends on the outcome of the integration of signals from multiple activated “pathways”. This is illustrated in the figure. Each specific signal binds to a specific receptor, where each receptor activates a particular signalling pathway : a cascade of activation events between different proteins (symbolized by different coloured circles connected by arrows). These events can end up in the cell nucleus where they may influence expression of certain genes.
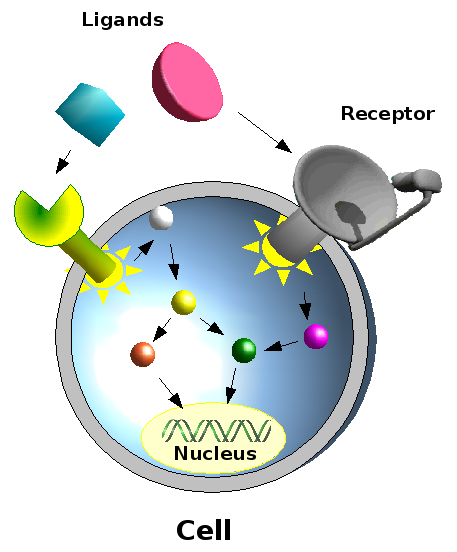 In our study we examined one of the
components in such a signalling pathway, the mitogen-activated
protein kinase pathway or MAPK pathway. This signalling pathway
is
involved in the every-day functioning of the cell, and when mistuned,
contributes to diseases like cancer and neurological diseases. By
studying these pathways we gain insight into how cells function
under
normal circumstances, and thereby understand more clearly what goes
wrong in diseased states. This allows us to develop measures to
intervene in such states.
In our study we examined one of the
components in such a signalling pathway, the mitogen-activated
protein kinase pathway or MAPK pathway. This signalling pathway
is
involved in the every-day functioning of the cell, and when mistuned,
contributes to diseases like cancer and neurological diseases. By
studying these pathways we gain insight into how cells function
under
normal circumstances, and thereby understand more clearly what goes
wrong in diseased states. This allows us to develop measures to
intervene in such states.
The focus of our study was the poorly characterized MAPK-activated protein kinase MK5. By means of cellular studies, we found out that MK5 cooperates with another signalling pathway component, namely protein kinase A or PKA, to induce changes in the F-actin cytoskeleton. These changes lie at the basis of cell motility and cell differentiation, which are often deregulated in e.g. cancer.
In another study, we developed a cell
line which expresses an overactive form of MK5 and
compared its
influence on gene expression levels. Besides describing a new way to
analyse these changes in gene expression, we found that MK5 changed
the expression of genes related to :
-
cancer phenotypes, confirmed by another group
-
movement processes e.g. development and/or differentiation, confirmed in our first paper and by others;
-
neurological processes, confirmed in our third paper.
In our third paper, we constructed a transgenic mouse line that expressed an overactive form of MK5. We saw that, compared to control animals, the transgenic female mice displayed less anxious behaviour while the transgenic male mice became more active. This indicates for the first time that a MAPK-activated protein kinase contributes to neurological processes.
Lastly, we also tried to determine which stimuli lead to altered MK5 expression levels. We found that stimulation by FSK could alter the mRNA levels of MK5, although this did not lead to increased protein levels. Furthermore, we also found that an element in the promoter of MK5, the cAMP Response Element, was responsible for basal transcription of MK5 because -compared to control cells- FSK stimulation did not lead to an increase in MK5 levels. Further studies are required to nail the exact regulatory mechanisms for MK5 expression.
In short, our research elucidated previously unknown roles for MK5 and indicates that it is an interesting player in several important cellular processes.





